Search78 Pictures
Results for Face
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 18738
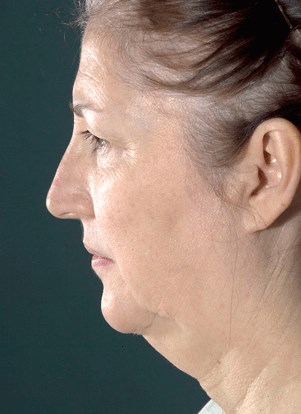
- Age:
- Over 60
- Gender:
- Female
- Ethnicity:
- Undisclosed
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 56348
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 17569
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10164
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10162
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10166
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 17549
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10168
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10169
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10154
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 17550
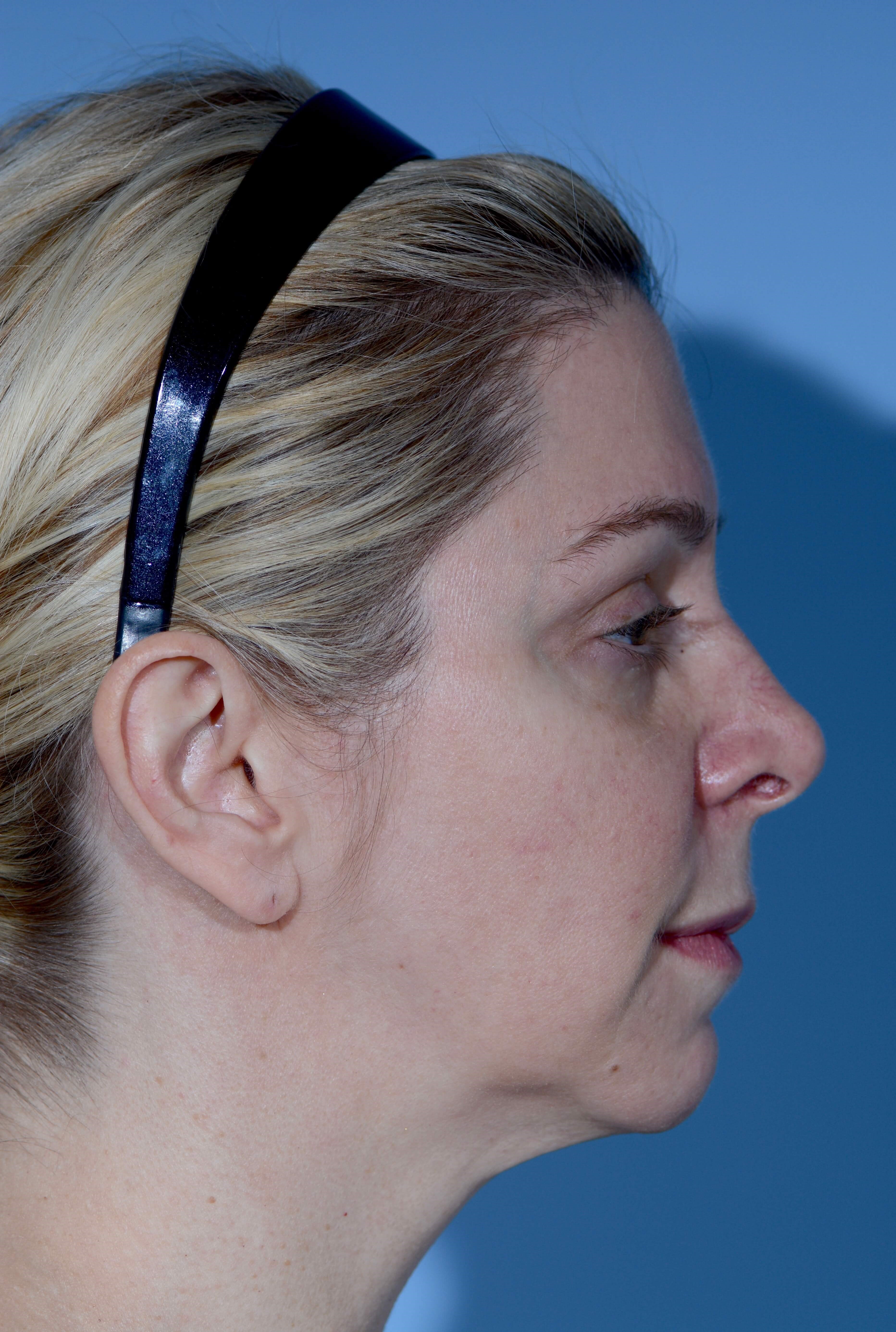
- Age:
- 40 - 49
- Gender:
- Female
- Ethnicity:
- Undisclosed
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 33088
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10162
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 18738
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10153
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10164
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10168
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10169
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 10154
- Height:
- Undisclosed
- Weight:
- Undisclosed
- Gallery:
- 17549
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)




&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)



&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)
&w=200&do-not-index)


